Transcranial magnetic stimulation, also known as TMS, has been providing relief to patients battling mental health or neurological conditions for years. Learn about the needs that led to the treatment’s development, the procedure itself and how it works, its eventual recognition by the FDA and more.

Transcranial magnetic stimulation (TMS) is a medical device treatment used to treat a number of different mental health and neurological disorders. Its technological apparatus sends out an electromagnetic field with the ability to safely reach brain structures found to take part in the appearance of the targeted condition. By influencing these structures, TMS is able to safely regulate their neural activity, creating a more stable cadence to their activation, thereby facilitating symptom alleviation and contributing to the patient’s well-being.
TMS treatment typically involves a series of 10-to-20-minute sessions over a period of several weeks. As it is noninvasive, TMS does not necessitate the use of anesthesia, nor does it require a lengthy recovery period. Patients are normally able to drive themselves to and from treatment sessions, allowing them to incorporate TMS into their daily schedules.
TMS does not cause any significant, long-lasting, or adverse side effects. Its most common side effect being a localized headache at the site of activation, which typically passes on its own after the first few sessions.
TMS therapy is offered by mental health clinics and professionals across the US and around the world, making the treatment more available than ever before. Its safety and efficacy have been recognized by research centers and national agencies across the globe.
TMS therapy was first made publicly available as a mental health treatment option during the 1980s. It was originally developed as an answer to the continued drawbacks of the available treatments at the time, namely: electroconvulsive therapy (ECT), psychopharmacology, and psychotherapy.
Electroconvulsive therapy, or ECT, is a medical device treatment that preceded TMS by several centuries. It sets off a series of brief seizures that stimulate the brain and affect its neural functioning.
ECT was originally used to treat schizophrenia, and in the 1960s-‘80s ECT was found to have greater efficacy as a treatment for mood disorders, particularly major depression. These days, it is considered one of the most effective treatments for depression and is mainly used to target this disorder.
Despite its proven efficacy, though, ECT does entail a number of drawbacks that prevent it from being considered a first-line mental health treatment for depression. Firstly, ECT can cause severe side effects, specifically short-term memory loss. It also requires the use of anesthesia, as well as a comparatively long recovery period, making it harder to work into one’s regular routine. Finally, due to widespread misinformation about the treatment, ECT is perceived as being a painful, traumatic form of therapy whose sessions leave patients incapacitated.
Due to its combination of high efficacy and often intolerable side effects, over the last few decades ECT has mainly been used in treatment-resistant cases of depression.
Psychotherapy, or talk therapy as it is sometimes called, is defined as a way to support individuals facing different mental health issues through communication. Usually, psychotherapy revolves around verbal communication, though certain types of therapy, such as play therapy for children, are also centered on other forms of activity.
Psychotherapy aims to help the patient better understand themselves, by examining key relationships, the patient’s self-perception, formative experiences, commonly used defense mechanisms, and other central aspects of their life.
Most types of therapy include the patient, therapist, and no one else. Additional therapeutic relationships can include couples’ therapy, group therapy, child-parent dyads, animal-assisted therapy and more.

Psychotherapy has been shown to be significantly beneficial to patients, with about 75% noting an improvement in their mental health. It can be experienced on its own, and can also be combined with other forms of treatment, such as psychopharmacology or medical device treatments.
Over the years, psychotherapy has evolved into different branches, each offering a different type of treatment, with its own advantages and disadvantages. Psychoanalysis, psychodynamics, and cognitive behavioral therapy (CBT) make up three of the most researched and well-known forms of psychotherapy today.
Conversation-based mental health support has existed throughout history, but it was first galvanized into its own form of therapy and profession toward the end of the 19th century, by the “Father of Psychoanalysis,” Sigmund Freud. Finding the reactions of his patients to defy conventional logic, Freud began exploring their pasts, connecting between events and their impressions left by them. Terms such as the consciousness, sex and death drives, defense mechanisms, melancholia and so much more of how humanity understands itself, the world and life were shaped by Freud, his successors and detractors, and as more perspectives were added to the discourse surrounding psychotherapy, so did new forms of therapy begin to appear.
Psychoanalysis is considered a rather intense form of therapy that is usually composed of three-to-five sessions a week. It can go on for years, as patient and therapist delve deep into the patient’s unconscious, in an attempt to gain a clearer understanding of how it works, while also developing adaptable defense mechanisms, thereby allowing the patient to, in Freud’s words, “work and love.”
Psychoanalysis has been found to be particularly effective with more engulfing mental health conditions, most notably with personality disorders. Cases of anxiety or depression have also been shown to benefit from the in-depth exploration that psychoanalysis can offer patients.
That said, the exhaustive nature of psychoanalysis can prove to be too adverse for patients to withstand, in addition to typically being very expensive. As a result, many do not continue with this treatment, or refrain from it entirely.
Psychodynamics
A form of psychotherapy that can offer short or long-term support, psychodynamic therapy is typically less intense than psychoanalysis, while still managing to offer patients a deep, introspective experience. Psychodynamic therapy usually involves one-to-two sessions per week and tends to emphasize the therapeutic relationship as a safe environment in which authentic change can manifest itself. Similarly to psychoanalysis, psychodynamic therapy has been shown to assist with personality disorders, and is considered a first-line treatment for depression.
However, psychodynamics still requires an openness on the side of the patient, as well as a willingness to be vulnerable and acknowledge experiences that can be painful to reconsider. Many patients therefore find this process to be daunting, and prefer more action-driven forms of therapy, or medicinal treatments, in lieu of psychodynamics.
This goal-oriented form of therapy is considered a first-line treatment for obsessive-compulsive disorder (OCD). As opposed to more immersive types of psychotherapy that focus on deep-rooted connections and relationships in one’s past, cognitive behavioral treatment (CBT) focuses on the patient’s present-day symptoms, in an attempt to bring about effective relief.
CBT has repeatedly been found to be an evidence-based treatment that targets adverse ramifications of mental health conditions and can provide significant improvement. That said, it is also very much reliant on the patient’s motivation. As a result, CBT may hit a difficult roadblock when faced with a patient’s internal resistance, preventing them from achieving true relief from their disorder.
As influential as psychotherapy can be, the therapeutic setting does not always feel comfortable to patients, who may struggle to tap into unsettling experiences or emotions that are linked to the mental health condition they are facing. With the advances of scientific mental health research, many individuals opt for a more empirical approach, preferring instead to try medication that has been recognized for their potential value in alleviating symptoms of mental health issues.

Research has aimed to offer scientifically backed solutions to the most pressing mental health issues of the day. As such, most mental health studies focus on one or more of the major mental health disorder families that cause significant harm to one’s well-being. These include:
Out of all of the above-mentioned families, anxiety and depression are considered the most common within the mental health world, with their symptoms regularly appearing in diagnoses. Certain scientific developments of the 20th century have addressed the concerns of anxiety and depression, by developing a group of medications commonly referred to as antidepressants—this despite all antidepressants also considered to be anti-anxiety medications, or anxiolytics.
Antidepressants influence the brain’s neural activity through a number of different pathways, reducing symptom prevalence and severity, while also stabilizing and elevating the patient’s mood. They are usually divided into three “generations” of medication, in addition to a fourth, atypical group:
The introduction of different psychopharmacological options helped many individuals struggling with mental health issues finally receive medicinal, empirically-based treatment that focused on the symptoms that most disturbed them. Nonetheless, even the researched and well-funded medication, such as antidepressants, are unable to offer significant relief to a large number of patients: out of those diagnosed with major depression, for instance, over 40% are found to be unresponsive to antidepressant medication. A similar percentage exists regarding obsessive-compulsive disorder (OCD), with studies finding that 40-50% of patients diagnosed with OCD to be considered treatment-resistant.
With such a large portion of patients found to be treatment-resistant, and adverse side effects continuing to serve as a deterring factor, additional, scientifically-based mental health treatment options were needed. For this reason, medical device treatments began to be studied, with transcranial magnetic stimulation being among them.

With a large portion of patients considered treatment-resistant, and the adverse side effects of ECT and certain mental health medications preventing many patients from seeking treatment, researchers, practitioners, and patients continued to look for cutting edge forms of therapy. And so, in 1985, a newly developed medical device treatment called transcranial magnetic stimulation (or TMS) was first made available to the public.
TMS was developed with the idea of improving upon its predecessors: unlike ECT, it does not cause significant, adverse, or long-lasting side effects. TMS therapy has also been shown to work particularly well in cases of treatment resistance when patients do not respond well to medication. And it offers an empirically-based, medical device treatment option that can appeal to those who do not find psychotherapy to be a sufficient form of support.
A noninvasive medical device treatment, TMS utilizes electromagnetic fields to safely regulate the neural activity of brain structures found to be associated with the targeted mental health condition. Two such conditions are depression, which has been associated with erratic activity in the bilateral prefrontal cortex and dorsolateral prefrontal cortex; and OCD, which has been associated with erratic activity in the anterior cingulate cortex.
An average TMS treatment session lasts between 20-40 minutes. During each session, the patient is seated in a comfortable chair, as the TMS operator focuses the machine’s electromagnetic fields on the targeted area of the brain. A full TMS treatment plan usually includes daily sessions over a four-to-five weeks, followed by periodic maintenance sessions.
By sending out pulses of electromagnetic fields, TMS therapy has been shown to create a more stable rhythm of activation within the targeted neural structures, thereby alleviating the frequency and severity of the patient’s presenting symptoms, facilitating relief, and improving their well-being.
In addition to TMS’s proven effectiveness as a standalone treatment, its noninvasive nature allows TMS therapy to be combined with other forms of treatment, such as psychopharmacology, which can improve the treatment regimen’s overall efficacy. Since it does not necessitate the use of anesthesia or a lengthy recovery period, TMS can be incorporated into the patient’s daily schedule, and allows for them to drive themselves to and from each treatment session.
At present, two types of TMS exist: Traditional TMS and Deep TMS™.
Traditional transcranial magnetic stimulation (traditional TMS), also known as repetitive transcranial magnetic stimulation (or rTMS), was the first type of TMS to be introduced on the market, in 1985. It has been recognized by the FDA to be a safe and effective treatment for the following mental health conditions:
Traditional TMS uses a figure-8-shaped, handheld device to send out its electromagnetic pulses, which manage to reach a brain depth of 0.27” (0.7cm), which has been shown to stimulate approximately 3cm3 of brain matter.
During a traditional TMS treatment session, the TMS operator holds the figure-8 over the targeted area, releasing the pulses to where the relevant neural structure is believed to be. Each traditional TMS therapy session takes about 40 minutes.
Though it has been found to be effective, traditional TMS treatments can run into targeting issues, with the use of a handheld device causing practitioners to miss the targeted brain structure. To avoid this, practitioners sometimes incorporate an additional, neuroimaging device that helps pinpoint the structure’s location.

Deep transcranial magnetic stimulation (Deep TMS) was made publicly available in 2013, as a development of standard TMS. It has been FDA-cleared to be a safe and effective treatment for the following mental health and neurological conditions:
Deep TMS uses its own, patented H-Coil technology to deliver electromagnetic fields deeper into the brain, reaching a significant sub-threshold of 1.25” (3.2cm), which has been shown to stimulate approximately 17cm3 of brain matter.
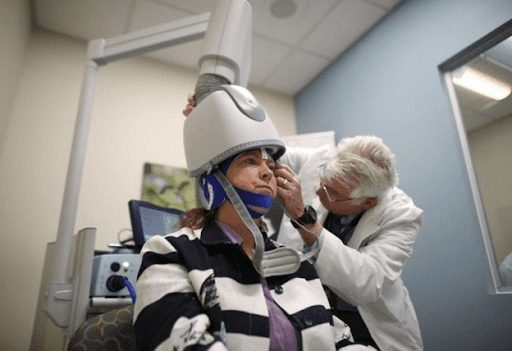
The H-Coil itself is held inside a cushioned helmet that is fitted onto the patient’s head. This allows Deep TMS’s H-Coil to reach wider swatches of the brain, while also directly reaching deeper neural structures. As a result, Deep TMS is able to avoid the targeting issues that sometimes arise with traditional TMS from occurring, resulting in increased efficacy. An independent* study published in 2019 in the Journal of Psychiatric Research seemed to confirm this, finding that patients with moderate-to-severe baseline depression experienced a significantly greater response level to a combined course of Deep TMS and medication, compared to either traditional TMS and medication, or medication alone.
A full-length Deep TMS treatment session lasts about 20 minutes, while the more recently developed, theta burst Deep TMS treatment for depression, lasts for three minutes. The treatment is divided into an acute phase that includes four weeks of daily sessions, and a continuation phase that includes two sessions a week over 8-12 weeks. Patients undergoing Deep TMS treatment often report experiencing a marked improvement of their well-being within three weeks of the initial session.
*The Magstim® Rapid² device was used in this study. (Magstim Company, Spring Gardens, UK)
As with other forms of mental health therapy, the main criteria by which TMS is evaluated are its efficacy and safety. It is by these standards that the probability of its success among patients seeking treatment is evaluated.
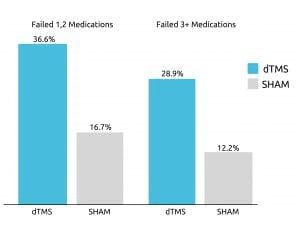
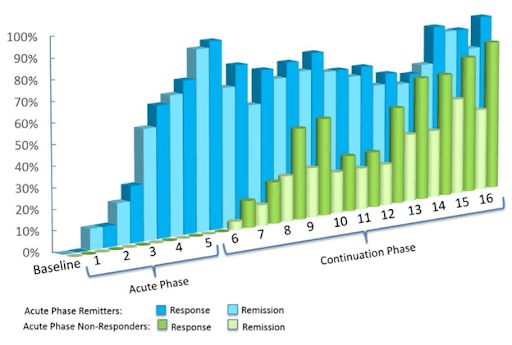
The efficacy of TMS has been repeatedly proven, with studies finding TMS to offer patients significant symptom relief. One such study, published in 2015 in World Psychiatry, examined Deep TMS’s efficacy with treatment-resistant patients of depression. The study relied on double-blind, randomized control trials, receiving data from 20 different research centers.
The study concluded that about one of three participants achieved remission after four weeks of Deep TMS therapy that is included in the treatment’s acute phase. The treatment was found to be successful even in cases where patients had found three or more types of antidepressants to be inefficient.
Additionally, over 80% of participants experienced an improvement during the treatment’s continuation phase.
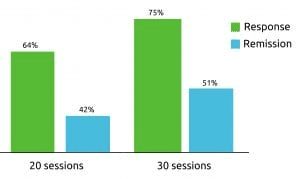
Deep TMS was reported to have even higher efficacy rates within a real-life clinical setting. with patients receiving the treatment in the field. Of the recorded data of more than 1000 patients undergoing Deep TMS therapy for depression, approximately 75% achieved a beneficial clinical response, and one of two patients achieved remission.
A different study on Deep TMS, published in 2019 in the American Journal of Psychiatry, focused on its efficacy in treating OCD. The study found that a six-week treatment course of Deep TMS provided patients with OCD significant relief that was similar to the results of a 12-week pharmacotherapy treatment. Over one in three patients experienced a significant decrease in OCD symptoms, including those previously determined to be unresponsive to medication treatment and/or psychotherapy. All in all, Deep TMS was concluded to be well-tolerated by participants, offering them the same level of symptom relief as medication, in half the time.
Deep TMS was also found to offer symptom relief when part of a treatment that combined it with medication. A study from 2019, published by the Journal of Psychiatric Research, concluded that undergoing Deep TMS together with standard antidepressant medication is significantly more effective than standard pharmacotherapy alone, as it reduced the symptoms of close to two-thirds of participants battling depression. Additional research similarly suggests that when treated with Deep and psychotherapy, the overall efficacy of the patient’s treatment plan will increase.
TMS therapy has repeatedly been shown to be a safe form of mental health treatment, beyond the relevant condition being treated. In a randomized, controlled study published in Clinical Neurophysiology, the safety of both traditional TMS and Deep TMS was found to be comparable to that of a sham group. Both types of TMS were shown to be well-tolerated, without causing participants any or long-lasting significant side effects.
Patients undergoing Deep TMS may experience some mild discomfort during the treatment session, in addition to a tapping sensation in the targeted area. In general, TMS side effects are usually quite mild.
The most common side effects observed following Deep TMS for depression are:
A double-blind, randomized, controlled, multi-center study on Deep TMS for OCD found no significant differences between the treatment and sham groups, suggesting these side effects may not be caused by the treatment itself. That said, the following, non-significant side effects were observed:
Patients with the following contraindications should refrain from Deep TMS treatment:
TMS therapy has been recognized by international agencies in the US and across the globe as a beneficial and well-tolerated mental health treatment option.
Due to a combination of safety and efficacy, both traditional and Deep TMS have been FDA-cleared to treat a number of mental health and neurological conditions.
Traditional TMS has been FDA-cleared to treat:
Deep TMS has been FDA-cleared to treat:
Deep TMS has also been recognized by the European CE certification mark, as a safe treatment option for a number of mental health and neurological conditions. The CE mark of approval states Deep TMS has met the health, safety, and environmental benchmarks of the products, services and devices that are sold within the European Economic Area (EEA).
As a result, Deep TMS has been CE-marked to treat:

Deep TMS for depression is covered by Medicare and most US private insurance providers, with OCD also now receiving wider insurance coverage. In many additional cases, single-pay coverage is also available. Patients with insurance coverage for Deep TMS usually incur the co-pay per treatment session stipulated by their insurance plan.
Deep TMS treatment is additionally covered throughout Europe and other regions that adhere to the European CE safety guidelines.