BrainsWay’s Deep Transcranial Magnetic Stimulation (Deep TMS™) therapy has been shown to have a high success rate in many clinical studies, which cited its ability to alleviate symptoms of a number of mental health conditions such as Major Depressive Disorder (MDD) and Obsessive-Compulsive Disorder (OCD). All studies cited in the following paragraphs rely on meticulous, evidence-based analysis whose results have been published in prestigious medical journals. They are based on gold-standard benchmarks, such as double-blind, randomized control trials (RCT) and multicenter patient arms to ensure a maximum level of objectivity.
A study from 2015 published in World Psychiatry, accumulated data from 20 different academic mental health centers that examine the effect Deep TMS had on treatment-resistant participants battling Major Depressive Disorder (MDD). The groundbreaking study, which was the foundation for BrainsWay receiving FDA-clearance for MDD treatment, found that roughly one out of three participants achieved remission after four weeks of Deep TMS treatment during the treatment’s acute phase. This was true even among patients who had previously failed to sufficiently improve from three or more antidepressant medications.
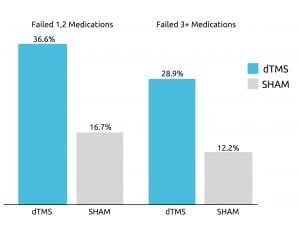
In addition, over 80% experienced a response (or improvement) during the following continuation phase.
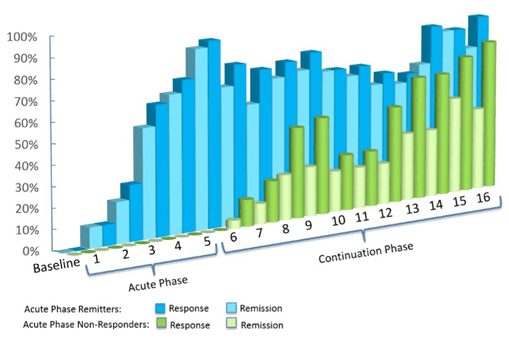
Deep TMS has been shown to have an even higher level of efficacy within real-life clinical settings, i.e. among patients receiving treatment in the field. Clinical data of over 1,300 patients in real practice settings who completed at least 30 sessions showed that approximately 4 in 5 achieved a response and 2 in 3 achieved remission.
BrainsWay’s Deep TMS treatment has also been clinically validated to treat OCD and has been FDA-cleared since 2018. Validating this stance is a study published in 2019 in the American Journal of Psychiatry, which found that a six-week course of BrainsWay’s Deep TMS treatment offered participants significant clinical improvement of OCD symptoms, with results similar to those of a 12-week pharmacotherapy treatment course.
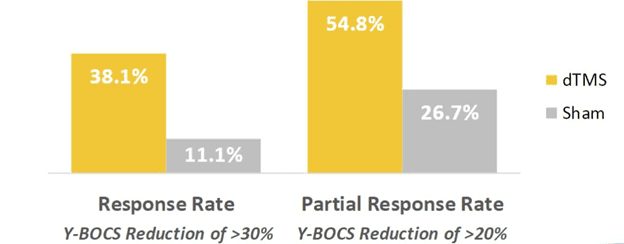
More than one in three patients achieved a significant reduction in OCD symptoms, including participants that were previously unresponsive to medication and/or psychotherapy. As a result, Deep TMS was cited as being a well-tolerated treatment with the ability to offer about the same level of relief as medication, but in half the time.
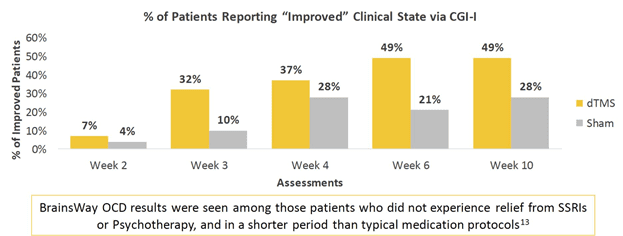
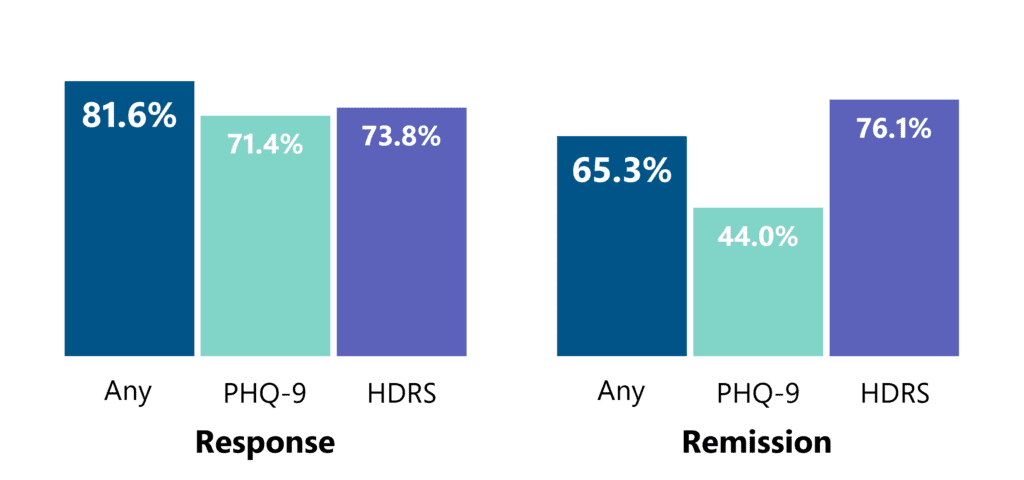
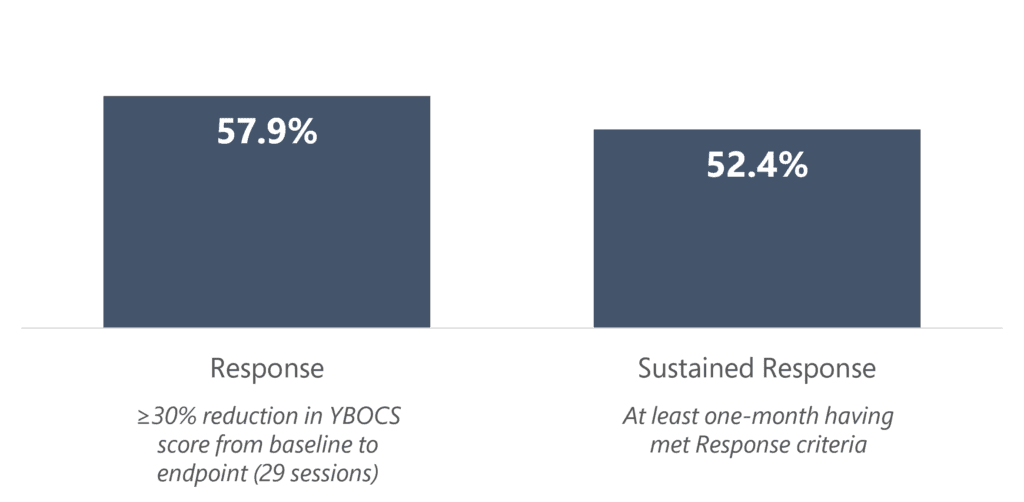
In real clinical usage of over 200 OCD patients, greater than 1 in 2 patients who received a treatment course of 29 sessions achieved response, indicating a 30+% reduction in symptoms as measured by the Y-BOCS scale.
It is safe to combine Deep TMS therapy with antidepressant medication or psychotherapy, as Deep TMS has also been found to offer relief when part of a combined treatment course. That said, a growing body of research has highlighted the treatment’s effectiveness in cases where people were found to be unresponsive to psychotherapy or SSRI medication.
In a study from 2019 published by the Journal of Psychiatric Research, Deep TMS therapy combined with standard medication for MDD was found to be significantly more effective than standard pharmacotherapy alone, reducing the symptoms of MDD in close to two-thirds of participants. These results emphasize Deep TMS’s ability to enhance existing treatment effects when provided in a professional medical setting.
Finally, many studies have suggested that TMS conducted during or in proximity to psychotherapy can increase the efficacy of both treatments’ combined effect.
With an increasing number of studies being carried out, Deep TMS therapy’s high level of efficacy is becoming more solidified, as it continues to improve the lives of many more individuals.
Read more about Deep TMS studies.