BrainsWay’s Deep Transcranial Magnetic Stimulation (Deep TMS™) is a patented, noninvasive treatment process that has been proven to alleviate symptoms of mental health conditions such as Major Depressive Disorder (MDD) and Obsessive-Compulsive Disorder (OCD).
During the Deep TMS treatment process, the patient is seated in a comfortable chair while a mental health practitioner fits a helmet apparatus holding BrainsWay’s patented H-coil technology onto the patient’s head. While the Deep TMS stimulator may be attached to separate treatment helmet simultaneously—for example, one geared toward treating MDD and the other for treating OCD—only one helmet and one patient can be activated at any given moment.
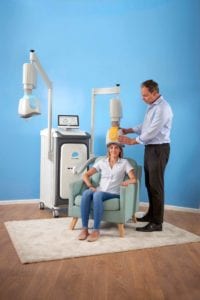
During the initial session, a mapping procedure will take place, involving single electromagnetic pulses delivered at different locations on the scalp, in order to determine the optimal treatment location and magnetic field strength. During all subsequent sessions, after making sure the patient is relaxed and the helmet is securely in place, the practitioner activates the Deep TMS device. The device then generates an electromagnetic field that safely stimulates brain regions found to be associated with the targeted mental health condition. The stimulation occurs for roughly 20 minutes, and may involve a brief verbal or visual exposure/provocation element to activate the neural pathways.
BrainsWay’s Deep TMS therapy has already been FDA-cleared in the US for both MDD and OCD treatment, and is CE Marked for additional psychiatric and neurological conditions in Europe. This innovative technology is an advancement of the standard TMS treatment, which was originally introduced in 1985 to help stabilize and regulate neural activity.
Deep TMS’s advantage lies in its ability to safely reach deeper areas of the brain directly, thereby managing to influence deeper brain structures without the need to increase the intensity of the electromagnetic field it utilizes. This ability to directly affect deeper brain structures also allows Deep TMS to broaden the scope of its stimulation, reaching more brain structures while keeping to a safe level of activation. The deeper and broader field of activation that Deep TMS offers increases its ability to reach and regulate the neural activity of more brain structures at the same time, while maintaining the procedure’s safety, and without relying on complex neuronavigation equipment.
As deep TMS continues to gain popularity within the mental health community, more and more patients are benefiting from the treatment’s effectiveness and noninvasive nature. Additionally, a growing number of mental health professionals are discovering how easy Deep TMS equipment is to operate, which allows them to confidently offer this treatment to their clientele.
With all these advantages, it is easy to see why Deep TMS is becoming a leading treatment option for mental health practitioners seeking to offer the most comprehensive care for those seeking symptom relief.
The Deep TMS device’s measurements and electrical information are as follows: