Journal: Frontiers in Neurology 11:584713 (2021)
Authors: F Spagnolo, M Fichera, R Chieffo, G Dalla Costa, M Pisa, MA Volonté, M Falautano, A Zangen, G Comi, L Leocani
Background:
Pilot open-label application of high-frequency Deep TMS in Parkinson’s Disease (PD) has shown promising results.
Objective:
To evaluate safety and efficacy of high-frequency Deep TMS in PD in a double-blind, placebo-controlled, randomized study.
Methods:
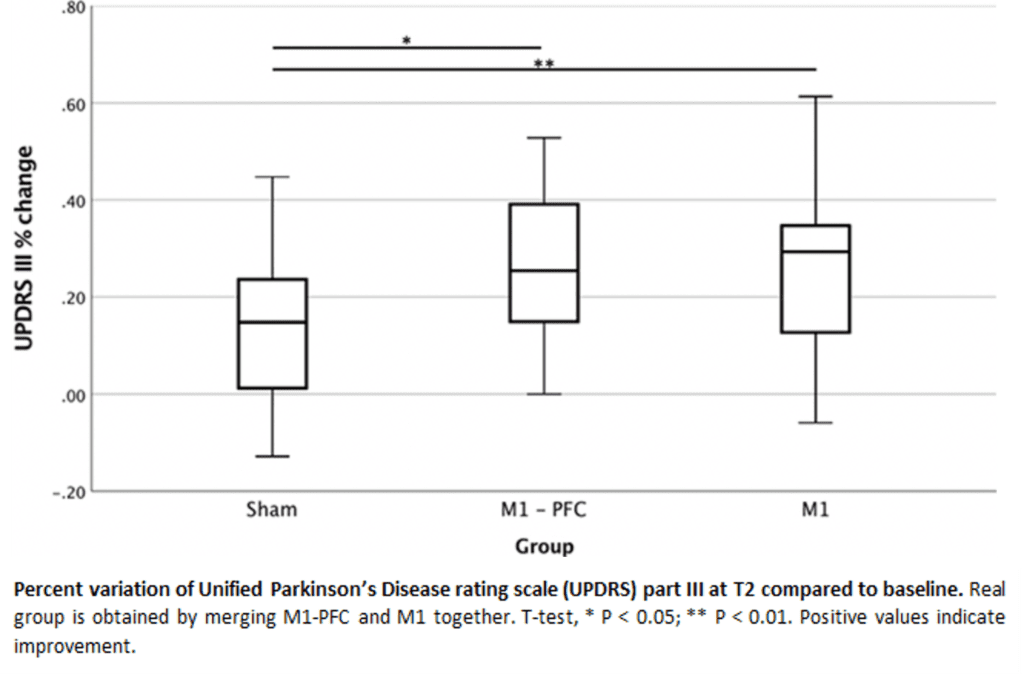
Sixty patients with PD were randomized into 3 groups: M1-PFC (real stimulation on primary motor-M1 and pre-frontal cortices-PFC), M1 (real stimulation on M1, sham on PFC), Sham (apparent stimulation). Primary outcome was baseline-normalized percent improvement in UPDRS part III OFF-therapy at the end of treatment (12 Deep TMS sessions, 4 weeks). Secondary outcomes were improvement in UPDRS part III sub-scores, timed tests, and neuropsychological tests. Statistical analysis compared improvement following real and sham stimulation at the end of the protocol using either a t-test or a Mann-Whitney test.
Results:
All patients tolerated the treatment and concluded the study. One patient from M1-PFC group was excluded from the analysis due to newly discovered uncontrolled diabetes mellitus. No serious adverse effect was recorded. At the end of treatment, patients receiving real Deep TMS (M1-PFC and M1 combined) showed significantly greater improvement compared to sham in UPDRS part III total score (p = 0.007), tremor sub–score (p = 0.011), and lateralized sub-scores (p = 0.042 for the more affected side; p = 0.012 for the less affected side). No significant differences have been observed in safety and efficacy outcomes between the two real Deep TMS groups. Notably, mild, not distressing, and transient dyskinesias occurred in 3 patients after real Deep TMS in OFF state.
Conclusions:
These findings suggest that high-frequency Deep TMS is a safe and potentially effective procedure and prompt larger studies for validation as add-on treatment in PD.