Roughly 17 million Americans struggle with depression. And yet the treatment options for depression are extremely limited, leaving millions of people to struggle with their symptoms. Current treatments, including antidepressant medication and psychotherapy, are insufficient and sometimes are even detrimental. Nearly 40% of people struggling with depression are resistant to treatment, while others experience the countless harmful side effects of antidepressants, or linger in psychotherapy sessions that have not been proven to show tangible results. Therefore, magnetic therapy is a true breakthrough in the treatment of mental illnesses.
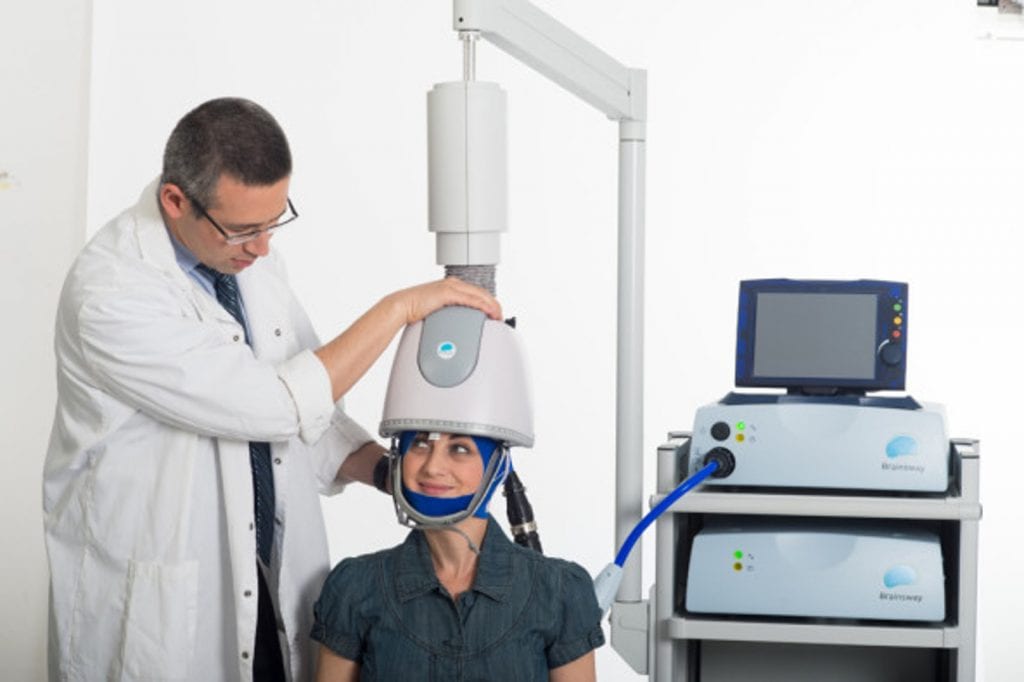
Transcranial Magnetic Stimulation, or TMS, is an innovative treatment for mental illnesses, including depression, that does not require any anesthesia or hospitalization. Magnetic therapy involves a helmet-like device, equipped with coils that generate a magnetic field to stimulate parts of the brain responsible for mood regulation. It is a noninvasive procedure that is well-tolerated and easily administered.
BrainsWay has developed a unique and patented coil system that enhances the magnetic stimulation and ensures that it reaches a deeper and broader brain region. BrainsWay’s Deep TMS is FDA cleared for the treatment of MDD and is the only TMS system that is also cleared for the treatment of OCD.

Magnetic therapy is a well-tolerated, noninvasive treatment for depression that stimulates parts of the brain. This treatment is FDA cleared and has been demonstrated to be effective at reducing or even eliminating symptoms of treatment-resistant depression. BrainsWay Deep TMS has zero systemic side effects, earning magnetic therapy its reputation for being a safe treatment option. Antidepressants produce countless harmful and debilitating side effects, however, TMS is a treatment option that has minor, nonsystemic side effects that amount to some patients reporting pain or discomfort in the jaw or at the application site. Adjustment of the helmet can reduce the pain or discomfort associated with treatment, which usually get better or go away completely with successive treatments.
BrainsWay Deep TMS helps provide people suffering from treatment-resistant depression and MDD with an alternative treatment option that only lasts five times a week for four-six weeks. BrainsWay prides itself on providing hope for a better future and a chance at achieving well-being.