Depression, or major depressive disorder, is a mental health condition that has been recognized throughout human history, dating back to the Ancient Greeks, through Freud’s definition of melancholia to more recent pharmacological developments that perceive depression as a neurological and chemical imbalance. These days, the American Psychiatric Association (APA) defines Depression combines elements of sadness, grief and pathological pervasiveness, resulting in a state of mind characterized by a deep unhappiness, low mood, lack of energy and a difficulty concentrating, over extended periods of time. Emptiness, loneliness, a lack of hope and an unrelenting sense of longing are all additional symptoms of major depression.
Major depression affects roughly one in 15 adults (or 6.7% of the overall adult population). In the US, 17.3 million adults (7.1%) reportedly have experienced at least one depressive episode over their lifetime. Up to 3% of children in the US are said to suffer from depression, as do up to 8% of US adolescents. Among the elderly, Depression also appears in 1%-5% of the elderly population, rising to 11.5% if they are hospitalized and 13.5% if they require home care.
Depression has been found in comorbidity with a number of other mental health conditions. These include obsessive-compulsive disorder (OCD), post-traumatic stress disorder (PTSD). It has also been linked to a significant increase in instances of self-harm and suicidality.
As a unique, non-invasive treatment, Deep TMS is able to provide relief to people who suffer from depression. And with the treatment now more readily available than ever, a growing number of patients are able to enjoy the benefits it provides.
Roughly 40% of people with depression are resistant to antidepressants. Deep TMS offers a solution to the many patients who find antidepressant medication to be ineffective, or too adverse in terms of its accompanying side effects.
By using a special, helmet-like device containing BrainsWay’s patented H-Coil, Deep TMS is able to offer an innovative way to treat depression. Over time, the Deep TMS treatment process is able to significantly diminish the symptoms of depression, providing the patient with the relief they had been searching for.
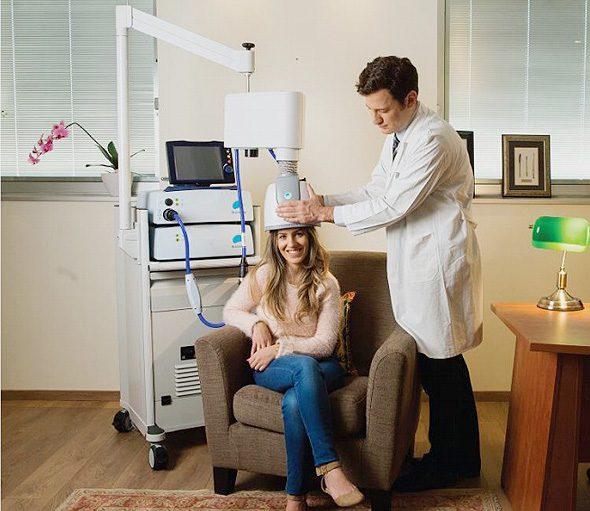
In 2009, Deep TMS was FDA-cleared as a treatment for Major Depressive Disorder (MDD). The clinical trials earning Deep TMS its FDA clearance status have shown BrainsWay’s Deep Transcranial Magnetic Stimulation to be a safe, well-tolerated, and effective treatment for depression. Research has shown that more than one third of medication-resistant patients achieved remission from MDD, thanks to Deep TMS. In real clinical practice, remission rates were even higher, with more than half of recipients achieving full remission.
Deep TMS is one of the few treatment options for depression that is both recognized by the FDA and has no significant, systemic side effects. A small portion of patients undergoing Deep TMS treatment report feeling limited side effects, including pain or discomfort at the application site or jaw. These side effects normally pass on their own, or following slight adjustments made to the helmet’s position on the patient’s head.
Deep TMS originally evolved out of First-Generation TMS, which uses a handheld, figure-8-shaped device to regulate neural activity. Deep TMS offers several advantages over its predecessor: while First-Generation TMS relies on a narrower scope of activation, Deep TMS’s patented H-Coil technology safely sends out its electromagnetic fields to wider swaths of the brain. This allows Deep TMS to avoid the targeting issues that sometimes arise during First-Generation TMS treatments.
Furthermore, Deep TMS is able to directly reach deeper brain regions, thereby regulating the neural activity of the structures found there. This direct regulation allows Deep TMS to provide a high level of efficacy when focusing on relevant neural structures that had previously been proven to be hard to access in a direct manner.