This Patient Manual is a supplement to the BrainsWay Deep TMSTM System Instructions for Use.
If you are considering BrainsWay Deep TMS treatment as an aid in short-term Smoking Cessation you should review this patient manual and discuss the information with your doctor in order to understand more about:
You are a chronic smoker (>10 cigarettes a day for more than 1 year) and your doctor has recommended treatment with the BrainsWay Deep TMS System. BrainsWay Deep TMS treatment has been shown to be safe and effective as a treatment aid for short-term smoking cessation in chronic smokers (>10 cigarettes a day for more than
1 year).
Your Healthcare Provider (psychiatrist or physician) will review your medical and smoking history to help determine if BrainsWay Deep TMS treatment is appropriate for you or not.
The treatment is performed under your healthcare provider’s supervision. Neither anesthesia nor sedation is required. You will be awake and alert during the treatment sessions. The treatment course consists of 5 daily treatment sessions per week for a period of 3 weeks, followed by once a week for another 3 weeks. Each treatment session lasts approximately 20-30 minutes. Prior to each treatment session, you will be asked to hold and handle a cigarette (from the brand you smoke) and a lighter. You will be asked to imagine that you are in a situation where you would normally smoke. Then Deep TMS stimulation will begin.
Deep TMS stands for “Deep Transcranial Magnetic Stimulation”. Deep TMS is a noninvasive procedure, involving stimulation of parts of the brain called the prefrontal cortex and insula. The stimulation activates nerve cells in these regions of the brain and assists in smoking cessation.
The stimulation is produced by a treatment “coil” contained in a helmet. During treatment, the coil is gently positioned on the frontal side of your head, over this region of the brain. Short pulses of electricity sent through the treatment coil generate magnetic fields that turn on and off very rapidly. These magnetic fields are similar to those used in magnetic resonance imaging (MRI) systems. The magnetic field goes through the head and induces a weak electrical current that briefly activates the nerve cells in this region of the brain and assist in short-term smoking cessation.
The BrainsWay Deep TMS System is indicated for use in adults as an aid in short-term smoking cessation.
Deep TMS treatment delivers a magnetic field that could cause any metal objects that are near the device to move or to get hot. Deep TMS treatment should not be used if you have metal implants in or around your head (except for standard amalgam dental fillings). Deep TMS treatment should not be used if you have implanted electronic devices in your head. These implants could cause serious injury or death if Deep TMS treatment is used. You should tell your healthcare provider if you have any metal devices or objects in your head or body in order to determine if those devices could be affected by the treatment.
Safety of the BrainsWay Deep TMS System was demonstrated in a clinical study involving 262 subjects defined as chronic smokers (>10 cigarettes a day for more than 1 year). The subjects ranged in age from 22 to 68 years.
This section summarizes the adverse events and side effects reported in the clinical study with the BrainsWay Deep TMS System
You should discuss the warnings and precautions related to the Deep TMS treatment with your healthcare provider to determine if any precautions should be taken prior to or during your treatment with the BrainsWay Deep TMS System.
There were no cases of a known seizure (also called convulsions) reported in the clinical study with the BrainsWay Deep TMS System. Two people did have twitching, which may or may not have represented focal seizure activity. In previous studies with the BrainsWay Deep TMS System, seizures have been reported in subjects on high doses of psychotropic medications, which are known to increase the risk of a seizure, due to high alcohol consumption the night before treatment, or due to other reasons. None of the subjects who have experienced Deep TMS-induced seizures have suffered lasting physical sequelae.
You should discuss with your doctor if you have had a seizure, or if you have a medical condition or change in a medical condition which may put you at increased risk of having a seizure, e.g., brain injury, change in medications, change in electrolyte balance, etc. Your doctor will decide if it is appropriate for you to receive BrainsWay Deep TMS treatment. Note that some medications can increase the risk of seizures, including those that may be used to aid in short-term smoking cessation. It is important to tell your doctor every medication that you are taking.
BrainsWay Deep TMS treatment may require several weeks of treatment before you begin to stop smoking.
You should also inform your doctor if you have any behavioral changes, such as increased aggressiveness, euphoria, or irritability. Your family should also be aware that if such behavioral changes appear, they should inform the treatment administrator immediately. Your doctor will determine whether BrainsWay Deep TMS treatment should be discontinued and, if so, what other treatment options are available.
During the course of 6 weeks of ongoing treatment, the treatment was safely tolerated by patients in the study. Furthermore, no negative effects of treatment were seen during the study nor in follow up 4 months after treatment. Longer term effects of exposure to the Deep TMS treatment are not known.
The safety and effectiveness of the BrainsWay Deep TMS System has not been established in the following patient populations or clinical conditions through a controlled clinical trial.
During treatment with the Deep TMS System a loud clicking sound is emitted. Patients must use earplugs with a rating of at least 30 dB of noise reduction. There have been no reports of hearing loss with the Deep TMS treatment in the clinical study when earplugs were used.
Table 1 below presents the adverse events reported in the clinical study in ≥5% or more of the patients who received the BrainsWay Deep TMS treatment or the sham (placebo) treatment. Safety information is provided from all patients who were treated in the clinical study.
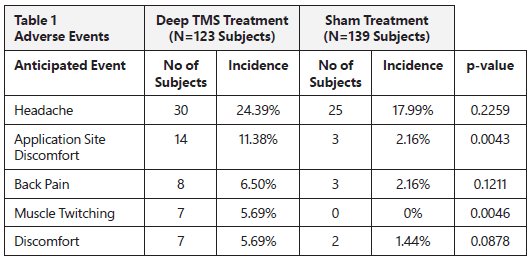
Headaches were reported in 24% of patients who received BrainsWay Deep TMS treatment, and in 18% of the patients who received sham (placebo) treatment. This indicates that headaches were not necessarily caused by the BrainsWay Deep TMS treatment. Headaches usually get better or go away altogether with successive treatments. Headaches may also be relieved by using common over-the-counter pain medications such as acetaminophen.
The differences in the reported adverse events in patients who received the BrainsWay Deep TMS treatment compared to patients who received the sham treatment that were statistically significant, were application site discomfort and muscle twitching.
You should Inform the treatment administrator if you feel pain or discomfort during the treatment. The Deep TMS helmet may be slightly adjusted on your head to relieve the pain or discomfort. Pain and discomfort associated with treatment usually gets better or goes away altogether with successive treatments.
Other side effects which may occur but are not necessarily caused by the BrainsWay Deep TMS treatment include pain in jaw or teeth and neck pain.
The safety and effectiveness of the BrainsWay Deep TMS System as an aid in short-term Smoking Cessation was demonstrated in a prospective, double blind, randomized, controlled, multicenter trial. The study was conducted at 14 study sites in the United States (12 sites) and Israel (2 site). During the treatment phase, TMS sessions were performed daily for 3 weeks, followed by one weekly treatment for another 3 weeks.
The primary endpoint was four-week continuous quit rate (CQR), representing abstinence during a consecutive 4-week period, until the 4-month follow-up. The 4-week CQR was statistically significantly higher (p-value=0.0238) in the Deep TMS arm (17.1%) than in the sham arm (7.9%). BrainsWay Deep TMS has a positive treatment outcome and has demonstrated a beneficial effect in short-term smoking cessation. Starting from week 2, i.e., from the target quit date (day 7-14), the number of cigarettes smoked was statistically significantly lower in the Deep TMS treatment arm compared to the sham arm, for all analysis sets.
You may ask your healthcare provider for more information regarding the clinical study.
The BrainsWay Deep TMS System has been shown to be safe and effective when used as an aid in short-term smoking cessation. You should discuss the treatment with your healthcare provider to ensure that BrainsWay Deep TMS treatment is the right treatment for you.
Most patients who benefit from BrainsWay Deep TMS began to experience results by the second week of treatment. The BrainsWay Deep TMS treatment should be administered daily for three weeks, followed by once a week for another three weeks. This is the treatment schedule that has been demonstrated as safe and effective in the clinical study.
As with any smoking cessation treatment, there is a risk that you may not stop smoking during the BrainsWay Deep TMS treatment.